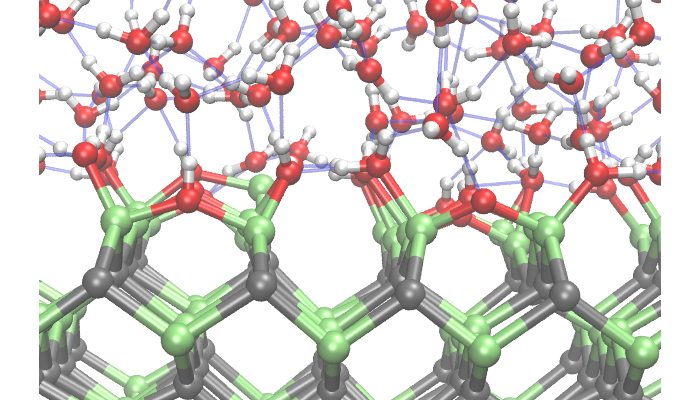
Lawrence Livermore scientists have developed methods and tools to predict the early stages of chemical degradation, oxidation, and corrosion of energy materials, and have applied the models to hydrogen production and storage.
Modeling technology performance
Among the most significant challenges for new and emerging energy technologies is the preservation of performance upon repeated cycling under real-world conditions. Chemical degradation is a key issue in materials for electrical energy storage, hydrogen storage, wind turbines, and catalysts for energy conversion. Likewise, preventing oxidation and corrosion of metals is critical for maintaining a reliable and robust energy delivery infrastructure.
We are developing tools to couple modeling and simulation at multiple scales to predict the early stages of chemical degradation, oxidation, and corrosion. These are combined with advanced in situ spectroscopy to assess dominant mechanisms and devise strategies for improving the lifetime of materials for next-generation energy production, storage, and delivery.
We have worked with partners at global universities and other national laboratories to develop and apply novel methods for simulating chemical reactions at interfaces, including oxidation and other undesirable reactions that limit the lifetime of energy-related materials. We use multiscale reaction models to couple these chemical reactions to solid-state diffusion and phase nucleation processes, which together determine the rate of early-stage degradation. Our protocols for tightly integrating simulation results with experimental microscopy and spectroscopy investigations of materials degradation leverage expertise throughout the Department of Energy (DOE) national laboratory complex.
Real-world applications
We have applied our models to predict and mitigate the chemical degradation of materials for low-temperature and high-temperature hydrogen production as well as candidate materials for solid-state hydrogen storage. Our reaction models also contribute to efforts within the DOE eXtremeMAT National Laboratory Consortium (XMAT), which seeks to develop approaches for mitigating hot corrosion in metal alloys for diverse energy-related applications, including engine and wind turbines and power generation.
The research is sponsored by the DOE Office of Fossil Energy, the DOE Fuel Cell Technologies Office within the Office of Energy Efficiency and Renewable Energy, and the DOE High Performance Computing for Energy Innovation program.
Publications
T.A. Pham, X. Zhang, B.C. Wood, D. Prendergast, S. Ptasinska, T. Ogitsu, Integrating ab initio simulations and x-ray photoelectron spectroscopy: Toward a realistic description of oxidized solid/liquid interfaces, J. Phys. Chem. Lett. 9, 194 (2018).
D.V. Esposito, J.B. Baxter, J. John, N.S. Lewis, T.P. Moffat, T. Ogitsu, G. O’Neil, T.A. Pham, A.A. Talin, J. Valazquez, B.C. Wood, Methods of photoelectrode characterization with high spatial and temporal resolution, Energy Environ. Sci. 8, 2863 (2015).
Y. An, B.C. Wood, J. Ye, Y.-M. Chiang, Y.M. Wang, M. Tang, H. Jiang, Mitigating mechanical failure of crystalline silicon electrodes for lithium batteries by morphological design, Phys. Chem. Chem. Phys. 17, 17718 (2015).
B.C. Wood, T. Ogitsu, E. Schwegler, Local structural models of complex oxygen- and hydroxyl-rich GaP/InP(001) surfaces, J. Chem. Phys. 136, 064705 (2012).
People
Learn more about this technology
- Looking to the sun to create hydrogen fuel (LLNL news, January 17, 2018)