Advanced manufactured materials developed at LLNL are well-suited for carbon capture, storage, transport, and delivery.
Carbon dioxide transport is difficult and expensive
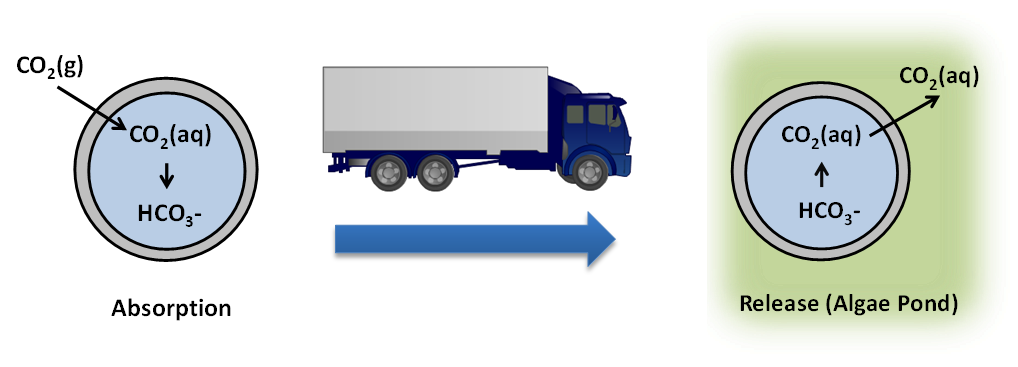
The ability to easily and cheaply transport carbon dioxide (CO2) from coal-fired power plants equipped with capture facilities to multiple, potentially distant utilization sites of widely varying scales will enable wider use of CO2 captured from flue gas. In the case of algal biomass cultivation, delivery of CO2 in the form of carbonate-based materials may provide a more efficient and economical method of inorganic carbon delivery than CO2 sparging, reducing the cost of algae cultivation and increasing the value of carbon capture.
LLNL innovations offer enhanced options
We have developed advanced manufactured materials that are well-suited for carbon capture, storage, transport, and delivery. These materials consist of aqueous sodium carbonate, which captures CO2 as sodium bicarbonate, extruded or 3D printed in a CO2 permeable polymer to increase the surface area and improve capture kinetics. By encapsulating carbonate solutions, LLNL has demonstrated order-of-magnitude enhancements in carbon capture rates compared with liquid carbonates, enabling the use of these (otherwise kinetically-limited) inexpensive, environmentally-benign solvents for carbon capture.
The use of carbonate materials for CO2 capture, transport, and delivery to algae has the potential to:
- Eliminate the requirement for co-location of algal production facilities with power plants or costly, low-volume pipelines.
- Develop a means of inorganic carbon transport, storage, and delivery tuned directly to seasonal and daily algal productivity level.
- Reduce capture costs by eliminating the need for CO2 stripping and compression processes.
We have demonstrated the biocompatibility of the materials with algal cultivation, the ability of the materials to load and deliver CO2 to algae over multiple cycles, and has scaled up synthesis by an order of magnitude.
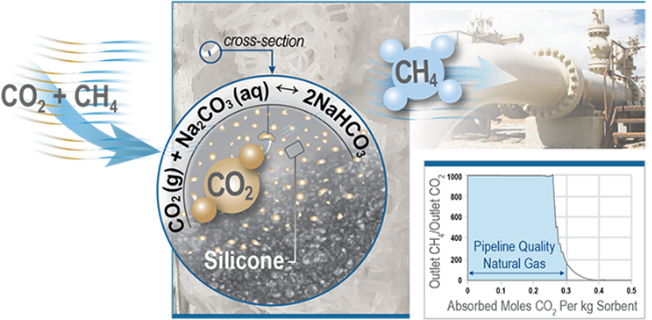
Additionally, we are developing cost-effective solutions for small-scale biogas upgrading—removing the CO2 so pure renewable gas can be injected into the natural grid—for biogas sources such as dairies and wastewater treatment plants. We demonstrated that carbonate composite materials offer an energy efficient solution for upgrading small-scale biogas streams. These materials can purify biogas from a wastewater plant to pipeline quality at ambient temperatures and pressures and are regenerated with a low energy input. We are working to demonstrate the scalability and stability of the materials at pilot scale.
For information about licensing LLNL carbon capture technology, please contact Annemarie Meike (meike1 [at] llnl.gov (meike1[at]llnl[dot]gov)).
Publications
M. Murialdo, H.M. Goldstein, J.K. Stolaroff, D.T. Nguyen, S.T. McCoy, W.L. Bourcier, M.R. Cerón, J.M. Knipe, M.A. Worthington, M.M. Smith, R.D. Aines, S.E. Baker, Three-Dimensional Printable Sodium Carbonate Composite Sorbents for Efficient Biogas Upgrading, Environ. Sci. Technol. 54, 6900 (2020).
D. Nguyen, M. Murialdo, K. Hornbostel, S. Pang, C. Ye, W. Smith, S. Baker, W. Bourcier, J. Knipe, R. Aines, J. Stolaroff, 3D Printed Polymer Composites for CO2 Capture, Ind. Eng. Chem. Res. 58, 22015 (2019).
K. Hornbostel, D. Nguyen, W. Bourcier, J. Knipe, M. Worthington, S. McCoy, J. Stolaroff, Packed and fluidized bed absorber modeling for carbon capture with micro-encapsulated sodium carbonate solution, Applied Energy 235, 1192 (2019).
J.K. Stolaroff, C. Ye, J.S. Oakdale, S.E. Baker, W.L. Smith, D.T. Nguyen, C.M. Spadaccini, R.D. Aines, Microencapsulation of advanced solvents for carbon capture, Faraday Discuss 192, 271 (2016).
J.J. Vericella, S.E. Baker, J.K. Stolaroff, E.B. Duoss, J.O. Hardin, J. Lewicki, E. Glogowski, W.C. Floyd, C.A. Valdez, W.L. Smith, J.H. Satcher, W.L. Bourcier, C.M. Spadaccini, J.A. Lewis, R.D. Aines, Encapsulated liquid sorbents for carbon dioxide capture, Nature Communications 6, 6124 (2015).
People
Learn more about this technology
- Art with a purpose: LLNL and startup company imagine the future of indoor carbon capture (LLNL news, May 23, 2019)
- Using microbes to convert CO2 to natural gas (, August 6, 2018)
- A Reversible Reaction Captures Carbon (Science & Technology Review, December 2017)
- Microcapsules capture carbon safely (LLNL news, February 5, 2015)